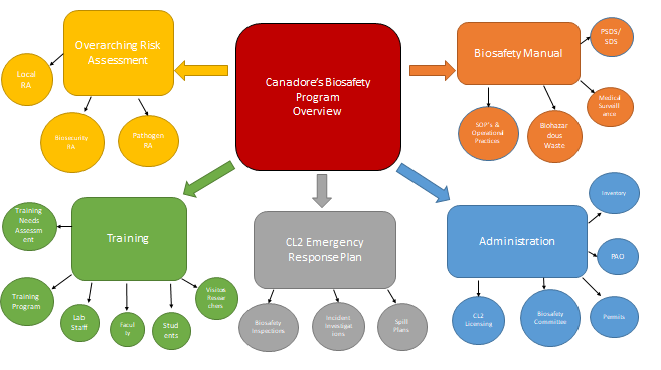
Canadian Biosafety StandardsCanadore College established its Biosafety Program in 2014 to comply with the Canadian Biosafety Standards, the Human Pathogens and Toxins Act (HPTA), and the Human Pathogens and Toxins Regulation (HPTR). It also supports other regulatory requirements as necessary. To ensure effective oversight, Canadore has created a Plan for Administrative Oversight (PAO) for managing pathogens and toxins in applied research within the college. This plan reflects a strong institutional commitment to overseeing the administration of the biosafety program. Biosafety and Biosecurity is addressed through a combination of policies and procedures, the biosafety manual and related SOP’s, risk assessments, training and administration.
|
Biosafety involves the principles, technologies, and practices designed to prevent accidental exposure to regulated materials and their unintended release. The Public Health Agency of Canada (PHAC) and the Canadian Food Inspection Agency (CFIA) assess the risk associated with pathogens and toxins to determine their Risk Group (RG). This classification helps establish the appropriate containment level needed for safe laboratory work and other activities involving these agents.
Risk Groups (RG) categorize biological agents (such as microorganisms, proteins, nucleic acids, or biological materials containing these elements) based on their characteristics, including pathogenicity, virulence, communicability, and the availability of effective preventive or therapeutic treatments. These classifications indicate the potential risk to human, animal, and public health, and are divided into four levels:
Risk Group 1 (RG1; Low individual and community risk): Includes microorganisms, nucleic acids, or proteins that either cannot cause disease in humans or animals or are unlikely to cause such disease.
Risk Group 2 (RG2; Moderate individual risk, low community risk): Consists of pathogens or toxins that present a moderate risk to individual or animal health but a low risk to public health and the broader animal population.
Risk Group 3 (RG3; High individual risk, low community risk): Includes pathogens that pose a high risk to the health of individuals or animals but a low risk to public health.
Risk Group 4 (RG4; High individual and community risk): Comprises pathogens that present a high risk to both individual and public health.
Canadore College has a Pathogens and Toxins License for a Containment Level 2: Laboratory Work Area for Risk Group 2 human pathogens and toxins and RG2 terrestrial animal pathogens and toxins. RG3 and 4 regulated materials are prohibited on campus.
Containment levels define the essential physical containment and procedural practices needed to handle regulated materials safely in laboratory, large-scale production, and animal research settings. There are four containment levels, ranging from the basic laboratory (CL1) to the highest level (CL4).
Containment Level 1 (CL1): This level allows for work with risk group 1 (RG1) materials, which can be conducted safely with standard microbiological procedures and basic physical containment measures.
Containment Level 2 (CL2): This level builds on the foundation set by CL1 and is suitable for working with a majority of RG2 materials. It enhances biosafety and biosecurity through more stringent operational practices and specific physical containment measures tailored to the increased risks associated with the materials handled.
The Canadore Genomics Research Hub operates as a combination of CL1 and CL2 physical and operational requirements depending on the activities taking place. In vitro work may be authorized for RG1 or RG2 regulated materials. In vivo work is prohibited at the college.
Canadore College offers a comprehensive Lab Safety Training Program for faculty, students, and researchers. This training includes general lab safety, biosafety protocols, the Public Health Agency of Canada’s e-Learning platform, internal training modules, biosafety manuals, standard operating procedures, and other relevant topics. The training is tailored to the specific materials and equipment you will encounter in the lab. For more details, please consult the training matrix.
Stakeholders wishing to utilize the laboratory space at Canadore College must adhere to all requirements outlined in the Biosafety Program and Plan for Administrative Oversight (PAO). This includes submitting a Canadore Biohazardous Materials Use Permit to the Biosafety Officer for review.
View the Research Project Flowchart
Canadore College is dedicated to continuously enhancing its Biosafety and Biosecurity program through an active institutional biosafety committee. This committee rigorously reviews all aspects of the Biosafety program and the work conducted within the labs to ensure ongoing improvements.
Helpful Resources
- Information on Biosafety and Biosecurity can be found on the Government of Canada’s website here
- PHAC’s eLearning Portal can be found here
- Information on Biohazard Containment and Safety under CFIA authority can be found here: Biohazard Containment and Safety - inspection.canada.ca
Contact Information
For more information about Biosafety and Biosecurity at Canadore College, reach out to the Science Lab and Biosafety Officer.
Corrie Duchesne, BSc., RBSO (CABS)
Science Lab and Biosafety Officer,
Schools of Environmental Studies & Health Science
Canadore College of Applied Arts & Technology
705.474.7600 ext. 5566
Corrie.Duchesne@canadorecollege.ca
100 College Drive, North Bay, Ontario P1B 8K9